Description
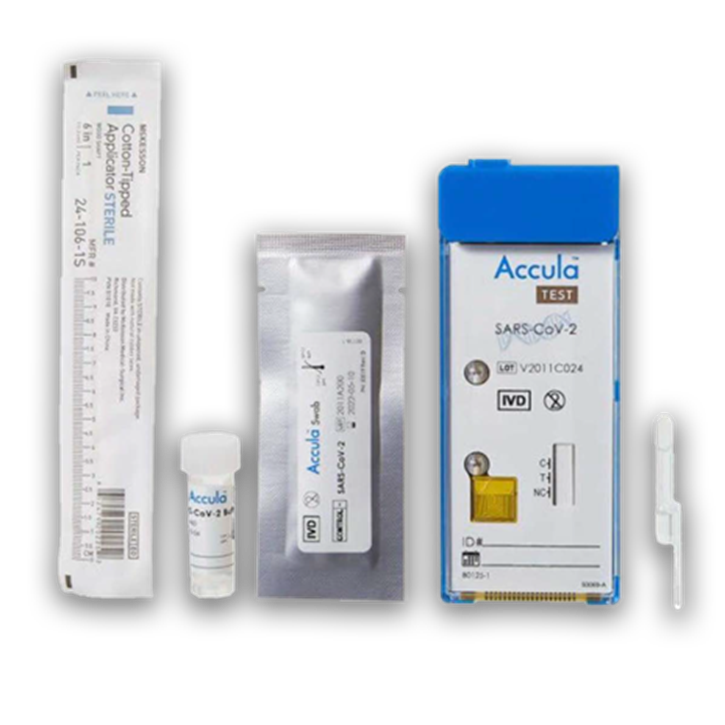
The Thermo Fisher Scientific™ Accula™ SARS-CoV-2 Test is a rapid diagnostic platform that combines the accuracy of RT-PCR with the simplicity, convenience, and procedural familiarity of traditional rapid immunoassays. The Accula SARS-CoV-2 Test has received an Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA) for the detection of SARS-CoV-2 in Clinical Laboratory Improvement Amendments (CLIA)–waived environments. Designed for use with the palm-sized Thermo Fisher Scientific™ Accula™ Dock, the Accula SARS-CoV-2 Test provides reliable, qualitative results in approximately 30 minutes.